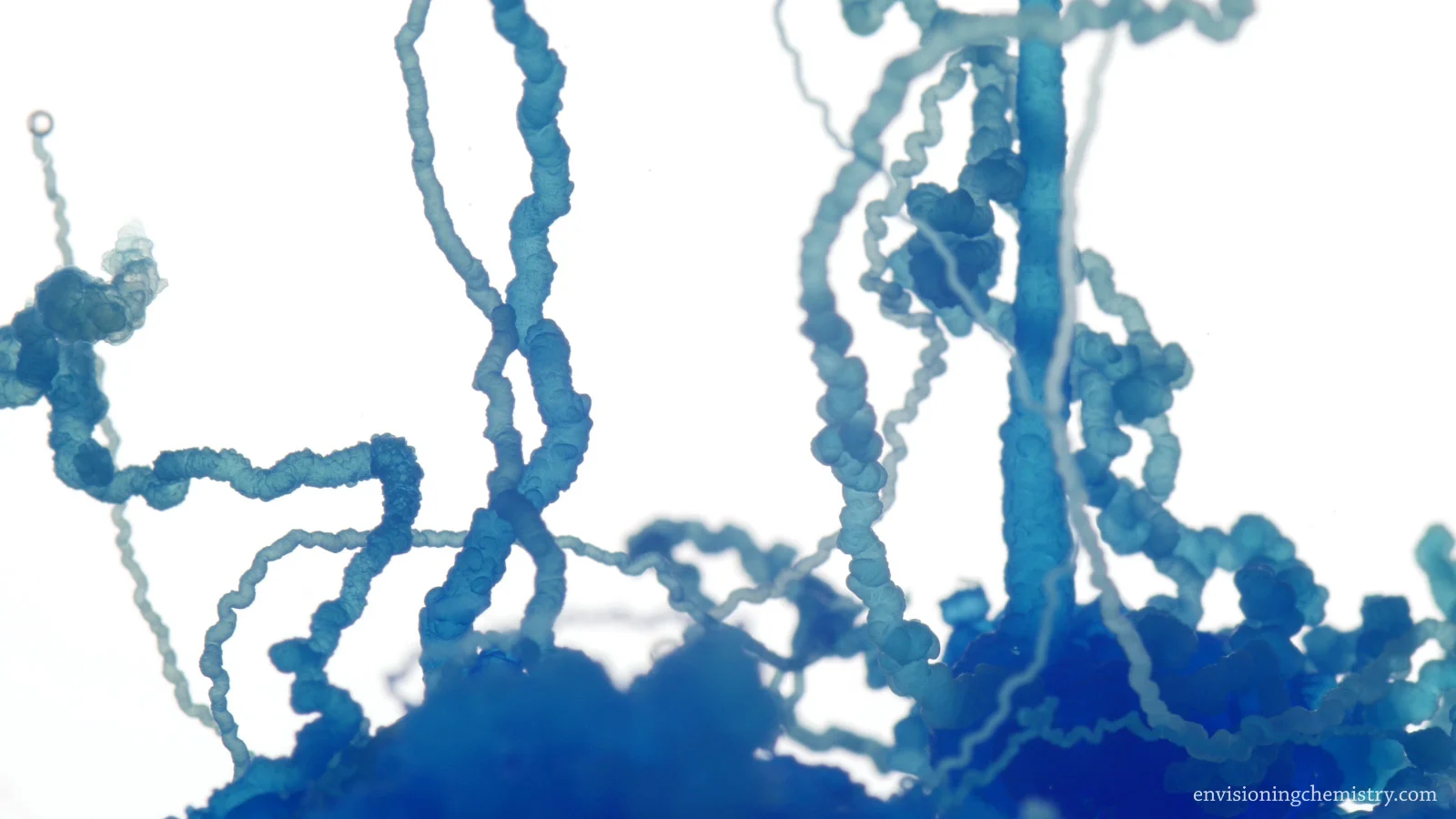
Chemical Garden II
When a piece of metal salt is dropped in the solution of sodium silicate, a membrane of insoluble metal silicate is formed. Due to the osmotic pressure, water enters the membrane and breaks it, generating more insoluble membranes. This cycle repeats and the salt grows into all kinds of interesting forms. This film recorded the osmotic growth of 6 salts inside sodium silicate solution. The growth was so life-like, no wonder Stéphane Leduc thought it might have something to do with the mechanism life over 100 years ago.
Cobalt(II) chloride in sodium silicate solution
Copper(II) nitrate in sodium silicate solution
Iron(III) chloride in sodium silicate solution
Copper(II) sulfate in sodium silicate solution
Zinc sulfate in sodium silicate solution
Calcium chloride in sodium silicate solution